Yes yes, I know. You’re right: it’s been far too long since I’ve posted. Well, I’m going to try to make it up to you with a nice article about one of the most influential and ubiquitous flavor components of beer: esters. I bet you’ve been waiting a long time for this article.
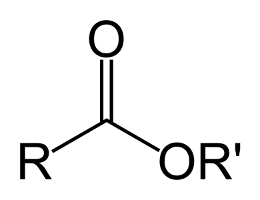
So, what are esters? If you ask a chemist they’ll tell you that esters are a class of molecules which contain a specific type of functional group called an ester group, if you can imagine that. These ester groups are made up of an oxygen molecule double-bonded to a carbon which is immediately adjacent to another oxygen which is bonded in-line with the carbon chain of the organic molecule. Perhaps a picture would illustrate the concept well.
Looking at that picture we see a portion of a larger molecule, where the R-groups represent what could essentially be any kind of organic chain. The ester group consists of the carbon and the two oxygens that are bound to it. Esters, due to the variation that can occur at those R-groups, are found in many shapes and sizes, but they all share the common feature of the ester group. The smaller weight esters are quite volatile and are frequently used in the production of food products and fragrances; they are largely responsible for much of the flavors and aromas associated with many types of fruits. Larger weight esters are also found everywhere, from DNA and plastics to triglycerides and explosives (nitroglycerin).
More after the break. Continue reading